|
What are The Three Types of Subatomic Particles?Ī typical atom consists of 3 types of subatomic particles: protons, neutrons, and electrons Protons 
If a neutron is added to the nucleus, it will produce an isotope of the same element, but if a proton is added to the nucleus (what happens inside the stars) it will be a completely new element. The number of protons is always the same as the number of neutrons inside a nucleus. 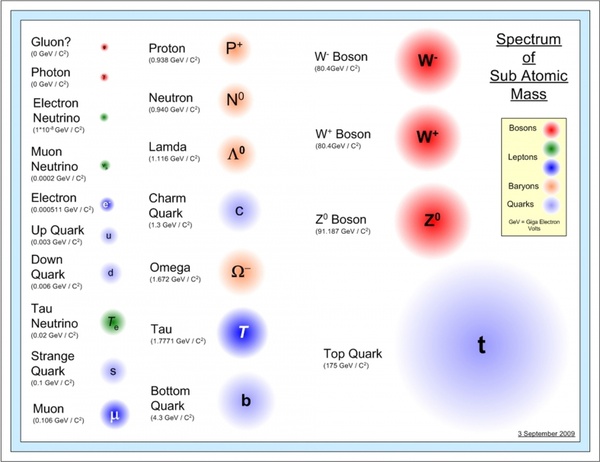
Inside the nucleus of the atom, we find different types of subatomic particles like protons and neutrons, which are heavier than electrons that rotate around the nucleus in a cloud that is 20,000 times the diameter of the nucleus. Visit Our Virtual labs and Experience a Virtual World of Science Education Try PraxiLabs Virtual Lab For FREE! This allows the protons to overcome the power of electrical repulsion, so they would not alienate from each other even though they have the same charge. Strong nuclear power (one of the four fundamental forces in nature) allows the components of the nucleus to hold together. The nucleus represents approximately the entire mass of the atom, where the mass of electrons is very small. He defined the positive particles inside the nucleus and called them protons, and he theoretically predicted the existence of particles with no electric charge, which was confirmed by his student James Chadwick in 1932 when he discovered neutrons. Rutherford was the first one to discover the nucleus more than 100 years ago. The process of the birth of stars, the composition of elements, the explosion of stars, and spread of elements in the universe still continue to this moment. These stars began to form heavy elements, then these elements had scattered throughout the universe because of the explosion of the stars at the end of their lives (supernovas). All of this happened during the first few minutes of the formation of the universe according to “CERN”.ģ80,000 years after the Big Bang, the temperature was adequate to slow the movement of electrons sufficiently to allow the nuclei to pick them up to form the first atoms in our universe – hydrogen and helium atoms – then, the forces of gravity were now able to perform their magic.īecause of gravity, the withdrawal of gas began to intensify (nebulas), which turned into giant factories of stars. Over time, quarks and leptons began to form protons, neutrons and electrons. We also know that they are made up of smaller particles such as quarks and leptons.Īfter the Big Bang, 13.7 billion years ago, our very hot universe began to expand and become cooler, allowing the formation of quarks and leptons. But now we know that atoms are made up of three types of particles: protons, neutrons, and electrons. The term “atom” comes from the Greek word Atomus, which means “indivisible.” At that time, the Greeks believed that the atom was the smallest thing in the universe and could not be divided. The atom is the smallest building stone or the smallest part of a chemical element that retains the chemical properties of that element, and thus it is considered as the basic unit of matter that controls the structure of the elements. Let’s start our journey from the atom to understand what it means. Other particles are produced from nuclear reactions, but they are unstable. Protons and neutrons are made up of smaller particles called quarks. Subatomic particles are particles much smaller than atoms, such as protons, electrons, and neutrons that make up the atom. The term particle always refers to objects of small sizes, and the most important types of particles are atomic and subatomic particles, each of which has different properties. In the physical sciences, a particle is a small localized object to which can be ascribed several physical or chemical properties, such as volume, density, or mass.  
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
